- (+33) 169867131
- neucrest@gmail.com

ESR1 - Subham SEAL
Institut Curie
I am Subham Seal, a PhD student working under the supervision of Dr. Anne-Hélène Monsoro-Burq in Institut Curie – Université Paris Saclay at Orsay. I am ESR1 (Early Scientific Researcher) for the Horizon 2020 ITN-NEUcrest program. I am of Indian origin and I did my Master’s at Tata Institute of Fundamental Research, Mumbai, India on the topic of cytokinesis in fission yeast.
I have always been interested in cell and developmental biology, especially in how cell and tissue migrations contribute to embryonic development. The complex, coordinated movements during gastrulation and neurulation fascinate me and as such, I am excited and look forward to working on the neural crest.
The neural crest is a multipotent tissue which gives rise to a host of different types of cells including neurons and glial cells, as well as bone, cartilage and melanocytes. Defects during the development of the neural crest may lead to multiple diseases such as Waardenburg syndrome and Hirschsprung disease. In the embryo, the neural crest arises from a tissue called the neural border, which forms between the axially positioned neural ectoderm and the lateral non-neural ectoderm. The neural border also gives rise to the sensory placode progenitors, a population of cells that give rise to the sensory placodes, and ultimately the different sense organs of the head and the cranial sensory ganglia. My project is focused on uncovering the genetic network that regulates the fate specification of the neural border into the neural crest and the placode progenitors. For this purpose, I am using the Xenopus laevis embryo as a model system.


ESR2 - Aleksandr KOTOV
Institut Curie
I am Aleksandr Kotov, a Ph.D. student in the Anne-Helene Monsoro-Burq research group at Curie Institute. I am a bioinformatician. My challenge in the Horizon 2020 ITN-NEUcrest program is to analyze the experimental data in model organisms and predict the neural crest gene regulatory network. I am Russian and have been working in oncology bioinformatics since 2016.
My background is a Bachelor’s degree in biotechnology and a Master’s degree in Science and Innovation Management. Apart from this, for more than 3 years I have worked as a bioinformatician developing a product for oncology therapy selection. Beginning from biotechnology research, I have always been interested in analyzing biological data using machine learning methods.
My research project is focused on constructing and understanding the gene regulatory network that determines early cell fate and behavior decisions in the ectoderm. This process results in the formation and migration of the neural crest, a crucial stem cell population for the vertebrate embryo. My research method includes bioinformatics analysis, machine learning approaches, and integration of transcriptome and epigenome data coming from diverse biological samples and species, generated by our team, as well as previously published research and from the human congenital disease database repositories. We expect to predict a genome-wide, multi-organism, and searchable NC-GRN focused on NC induction, EMT, and cell migration. The GRN will be validated in vivo experimentally (with help of ESRs 1&3).

ESR3 - Chloe PAKA
STEMCELL Technologies
Neuroblastoma is one of the most common cancers found in infants, accounting for up to 15% of all childhood cancer related deaths. It is thought to arise during development of the embryo from the neural crest, a cell type found in the embryo. These cells are responsible for the development of neurons and cells of the adrenal medulla, an endocrine gland found near the kidney. This system, known as the sympathetic nervous system, causes the "fight-or-flight" response, which allows our bodies to respond to stress. However, in neuroblastoma, the neural crest cells responsible for the development of the sympathetic nervous system do not differentiate properly during embryonic development, and form tumors, events that are more likely to occur when specific genetic mutations are found in the patient.
In this context, the aim of my PhD project, conducted both at STEMCELL Technologies and in Karen Liu's team at King's College London, is to understand the impact of genetic mutations linked to neuroblastoma on generation of the neural crest and the sympathetic nervous system. To this end, we will create a model of neural crest and sympathetic lineage differentiation in a dish using patient induced pluripotent stem cells (iPSCs). Human iPSCs are stem cells generated from any cell in the body, allowing us to study a patient’s own cells without any invasive procedure. This technique was awarded a Nobel prize in 2012. Using this tool, we will investigate neuroblastoma-related mutations in the anaplastic lymphoma kinase (ALK) gene, that is known to be involved in several other cancers and associated with poor prognosis in neuroblastoma patients. We hope this approach and associated findings will uncover new disease mechanisms which may contribute to the development of novel treatments and therapies for neuroblastoma.


ESR4 - Anna WULF
King's College London
I am Anna Wulf and I started my PhD in Karen Liu’s group at King’s College London in July 2020. I completed my bachelor’s and master’s degrees in biochemistry at the Freie Universität of Berlin, Germany. During my studies, I gained much practical and theoretical experience in biology, chemistry, and biochemistry. Furthermore, I identified my passion for science during self-organised research projects.
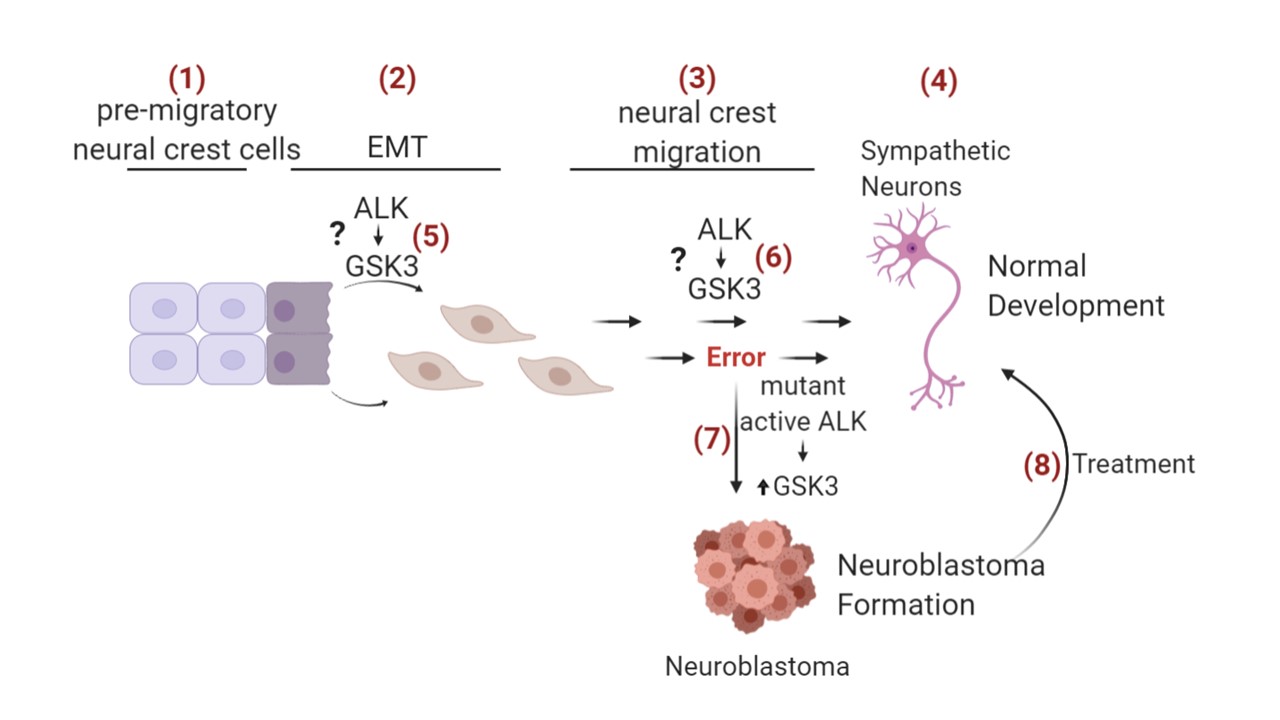
The aim of my PhD will be to identify key pathological events in the onset of neuroblastoma by using both in vitro and in vivo systems. Neuroblastoma is a neural crest cell derived cancer and is one of the most common and lethal childhood cancers. Neural crest cells derive from the neural plate (pre-migratory neural crest cells) (1), undergo epithelial–mesenchymal transition (EMT) (2), migrate throughout the embryo (3), and can differentiate into neurons (4), pigment cells, craniofacial cartilage and bone. Aggressive neuroblastoma has been associated with an increased activation of anaplastic lymphoma kinase (ALK) (e.g. activating mutation F1174L and R1275Q) and subsequent activation of glycogen synthase kinase 3 (GSK3) (7) but it is unclear how these kinases influence neural crest migration and differentiation. The focus of my thesis will be on understanding the pathological kinase activation in neuroblastoma in the context of changes in neural crest migration. ALK is a tyrosine kinase receptor and its activation is shown to increase GSK3 tyrosine phosphorylation on the leading edge of migratory neural crest cells (5). By using immunoprecipitation and immunofluorescence microscopy, I want to investigate whether GSK3 phosphorylation is caused by direct interaction with ALK or if other binding partner are necessary (indirect interaction). In addition, I will examine wildtype and mutated active ALK and its influence on GSK3 phosphorylation in context of neural crest migration and development (6). Another aim is the understanding of downstream targets of tyrosine phosphorylated GSK3 in neural crest cells, and how they differ in neuroblastoma cells. This will elucidate relevant cellular changes during initiation of metastasis. Investigating the normal and pathological migration of neural crest cells by modifying ALK and GSK3 signalling can help to find new drug targets for neuroblastoma treatment (8)

ESR5 - Amy KERR
University of East Anglia
My name is Amy, I am from Hamburg, Germany and during my studies at Maastricht University, The Netherlands and the University of Aberdeen in Scotland, I quickly became fascinated by developmental biology and how small errors in the complex mechanisms regulating embryonic development can have far reaching consequences. Therefore, I have joined Prof. Wheeler’s laboratory at the beginning of October 2020 to study the development of the adrenal gland and to identify associated neurocristopathies with this organ.
The adrenal gland is a key regulator in the human body, involved in the regulation of major physiological processes through their main function as a hormone supplier. The inner part of the organ, the adrenal medulla is derived of neural crest cells, the so-called chromaffin cells, that produce catecholamine hormones adrenaline and nor-adrenaline that regulate the 'fight-or-flight' stress response.
However, to this day, the embryology of this organ has not been well studied, making it difficult to assess underlying mechanisms involved in the onset of several disorders caused by adrenal dysfunction. Therefore, my role will be to characterize the development of the adrenal gland and identifying novel disease variants using Xenopus laevis as a model which could be used for potential drug discovery for treatment of human adrenal diseases.

Comparison between the human and frog adrenal gland structure and position. Thehuman adrenal gland sits on top of the kidney while the frog adrenal gland sits on the ventralsurface, however not much is known about the development and location of the adrenal medulla inthe frog.

ESR6 - Marco ANTONACI
University of East Anglia
Despite all our cells share the same genome, there are about two hundred different cell types in human body. What makes the difference between these cells is their genetic profile, or the way they regulate their genome. Micro RNAs (miRNAs) are short (20-22nt) RNA molecules capable to finely tune the expression of specific genes, thus they contribute to the diversity of gene expression between different cell types.
The Wheeler laboratory, at the University of East Anglia, focuses its research on the study of neural crest cells (NCCs) using the model organism Xenopus. Their goal is to gain insight to the mechanisms that give rise to neural crest-associated diseases, the so-called "neurocristopathies". In recent years, they have focused their attention on the role of miRNAs on early NC development.
In the Wheeler lab, two miRNAs have been identified as expressed in NCCs (miR-196a and miR-219). The role of these miRNAs is still to be elucidated, although preliminary studies show that both of them are important for the formation of neural crest tissue.
The role of the ESR 6 will be to find the target genes of these miRNAs and to enlighten their role in NC development. The success of this project will increase our knowledge of the gene regulatory network that underlies the formation of the NC tissue. More than this, it could give precious information about the molecular mechanisms of several neurocristopathies (such as the DiGeorge Syndrome). It will also help scientists who are working with human induced pluripotent stem cells-derived neural crest (hiPSCs-derived neural crest) to differentiate and characterize them.
In summary, this project will provide an important transverse help to the NEUcrest members, particularly to the ESRs that are working with gene finding and hiPSCs.


ESR7 - Haris Bin FIDA
National University of Ireland Galway
I am Haris Bin Fida, an early stage researcher (ESR) 7, currently working under the supervision of Dr. Gerhard Schlosser, School of Natural Sciences, National University of Ireland, Galway, Ireland. I am from Pakistan, and after finishing my Bachelors in Microbiology, I went to Belgium to pursue a Master of Science in Molecular Biology at the Katholieke Universiteit Leuven.
During my M.Sc. I got an opportunity to explore my research potential at the brain research group of the Belgian Nuclear Research Center (SCK-CEN). The objective of my research was to elucidate the role of p53 in radiation-induced microcephaly in mice. In this study, we dissected the cellular and molecular mechanisms underlying radiation-induced microcephaly. Performing this research acquainted me with sound practical and theoretical experience in molecular and cell biology, and fostered my interest in developmental biology.
My current PhD project focuses on elucidating neural crest (NC) specification in Xenopus laevis animal model and gaining more insight into the cellular and molecular mechanisms underlying the dysregulation of NC specification. The NC is one of the most intriguing cell populations of the developing vertebrate embryo. It gives rise to many anatomical features and cell types such as the craniofacial skeleton, neurons and glia of the peripheral nervous system, and melanocytes. The importance of NC development in humans can be exemplified by the fact that defects in its specification, differentiation, or migration result in various congenital disabilities known as neurocristopathies.
NC development is a multistep process, and it initiates during gastrulation from a broad territory located between the neural plate ectoderm and the non-neural ectoderm, termed the “neural plate border” (NB). The NB also gives rise to placode progenitors, which contribute to various cranial sense organs and nerve ganglia. My main objective is to identify and functionally analyze different candidate genes involved in the specification of cell fates at the NB, and elucidate their interaction with other transcription factors and signals constituting the gene regulatory network (GRN) at the NB. Deciphering the NB-GRN will reveal how cells become committed to either NC or placodal lineages at the NB. Ultimately, investigating the NC specification at the molecular level will give more insight into the etiology of various neurocristopathies and their better therapeutic management.

ESR8 - Merin LAWRENCE
National University of Ireland Galway
I am Merin Lawrence and I am doing my PhD at National University of Ireland in Dr. Gerhard Schlosser’s lab. I completed my bachelors in biotechnology from India and my masters in molecular medicine from United Kingdom.
Neural crest and placodes present shared aspects of development. Both are embryonic tissues that give rise to multiple non-epidermal cell types including neurons, glia, and secretory cells. These cells migrate and differentiate to multiple cell types whilst the tissue develops. Finally, both neural crest and placodes develop from populations of cells near the border of the neural plate. Sox9 and Sox10 are members of the Sox E family of transcriptional factors and are important contributors to neural crest and otic placode development. They are also implicated in several neurocristopathies. Despite having an essential role in embryonic development, the mechanisms behind the functioning of these transcription factors during neural crest and otic placode development is poorly understood. The given EU Horizon 2020 ITN-Neucrest project aims to investigate the role of SOX9 and SOX10 in neural crest and otic placode development. Target genes of SOX9 and SOX10 that are specific to neural crest and otic placode development will be investigated through RNA-seq and Chip-seq analysis. In addition, explants will also be analysed after knockdown of SOX9 and SOX10 genes. Subsequently, knockdown studies will be performed for some of the target genes using gene-editing techniques in Xenopus and hiPSCs. The data thus generated will be used for genetic functional analysis. The expected results will provide valuable insight into the role of SOX9 and SOX10 in Xenopus neural crest and otic placode development.


ESR9 - Sanjay VASUDEVAN
Institute of Neurosciences, CSIC and UMH, Spain
I’m Sanjay Vasudevan, carrying out my PhD under the supervision of Prof. M. Angela Nieto at the Institute of Neurosciences, CSIC and University Miguel-Hernández (UMH), Alicante, Spain.
I did my Bachelor of Technology in Biotechnology in India and later on moved to Sweden to carry out my Master of Science in Cell and Molecular Biology at Uppsala University. I have spent most of my time in academic research studying the potential use of stem cells in various regenerative medicine applications and disease modelling of developmental disorders. I initially worked on bone regeneration using mouse mesenchymal stem cells during my bachelor education in India and later on started working on neurological disease modelling using induced human Pluripotent Stem Cell derived neural crest cells during my master’s at Uppsala University. I believe that passion and purpose are extremely important in science and hence have always directed my research career in such a way that all my previous research works contribute towards my next step in a streamlined manner and that’s exactly why I believed that this NEUcrest ITN was just ideal to assist my transition into a PhD candidate in the field I love the most.
Epithelial to mesenchymal transition (EMT) is a term that I constantly came across throughout my academic research due to its importance in development and cancer. The opportunity to carry out my PhD under the supervision of one of the pioneer’s in the field in Prof. Angela Nieto is something that I grabbed onto in a flash.
During early developmental stages of an embryo, folding of the neural plate (neurulation) induces formation of neural crest cells (NCCs) along the border between the ectoderm and neuroepithelium. NCCs are multipotent cells with high migratory characteristics. They contribute to the formation of a wide range of cell types such as neurons and glia of the peripheral nervous system, sympathoadrenal cells, cranial bone and cartilage, smooth muscle cells and melanocytes. Flawed or pathologically regulated EMT-TF during NCC development can potentially lead to a broad range of disorders and congenital malformations referred to as neurocristopathies, or tumors derived from the neural crest. NCCs acquire the ability to delaminate and migrate in response to environmental cues through the activation of the Epithelial to mesenchymal transition (EMT) programme. Coordinated activity of a plethora of EMT transcription factors (Snail, Prrx, Twist, Zeb gene families) induces major changes in cell junctions, adhesion properties and cause cytoskeletal rearrangements. Preliminary data from the laboratory indicate that when epithelial cells express these factors individually, they all undergo EMT, but their migratory behaviour is different, which explains why the organism needs so many different factors. The hypothesis of my project is that the combinatorial expression of the EMT-TFs gives rise to different behaviours of the migratory cells that can be correlated with the relative position and the destinies of the different populations of the neural crest. Therefore, it is crucial to decipher the molecular basis of neural crest cell behaviour during EMT across multiple model systems.

ESR10 - Nitin NARWADE
Institute of Neurosciences, CSIC and UMH, Spain
I am Nitin Narwade, working as a Ph.D. student under the supervision of Prof. Angela M. Nieto at the Institute of Neurosciences, CSIC and University Miguel-Hernández (UMH), Alicante, Spain. I did my bachelor’s and master’s in bioinformatics in India and I have over 3 years of experience as a professional Bioinformatician involved in various aspects of genomics data analysis including cancer genomics and database or web server development.
While working as a cancer genomics data analyst, I came across the term Epithelial to Mesenchymal Transition (EMT) through a literature survey which immediately caught my attention. Initially, I started exploring EMT in the context of cancer metastasis, and eventually, with my thriving interest, I started exploring EMT in terms of embryonic development. The EMT is a molecular process that implies the conversion of an epithelial cell to a mesenchymal cell which adopts the migratory property. Prof. Angela Nieto’s lab is one of the leading research groups working on EMT for the last 20 years and continuously contributing towards its better understanding in different contexts including development and disease.
I joined the NEUcrest project intending to investigate and better understand the process of EMT during normal and defective Neural Crest (NC) development. The NC cells are the highly plastic multipotent embryonic stem cell population that has unique migratory properties. NC cells delaminate from the neural tube during embryonic development and start migrating to the target sites where they give rise to distinct cell derivatives such as melanocytes, chondroblasts, neurons, glia, etc. The abnormal specification, migration, differentiation of NC progenitor leads to severe abnormalities and birth defects such as cleft palate disorders, craniofacial and heart anomalies, hearing disorders, NC derived cancers (neuroblastoma, melanoma, etc.).
In my project, I am using a genomics-based approach to identify enhancers together with transcriptomic analysis to study the EMT gene regulatory landscape during NC development. I am also aiming to do the comparative analysis of normal and defective NC development in different vertebrates which helps us to better understand the common and organism-specific EMT program during NC development. The comparative study between normal and defective NC will allow us to predict the potential molecular targets for neurocristopathies.


ESR11 - Filipa DUARTE
Erasmus Medical Center, Rotterdam
I am Filipa Duarte, working at the Erasmus Medical Center in Rotterdam, the Netherlands. The ESR11 project arises from a collaboration between the Department of Plastic and Reconstructive surgery and the Department of Calcium and Bone Metabolism from Erasmus MC. Additionally, it counts with the participation of the Department of Craniofacial Development from King's College of London.
Craniosynostosis is a condition characterized by the premature fusion of one, or, several cranial sutures. It leads to facial deformities that frequently affect vision, breathing, dentition, hearing loss and cognitive impairment resulting from raised intracranial pressure. Neural crest cells are formed during development. Cells from the cranial neural crest migrate to give rise to the craniofacial mesenchyme and generate the majority of cranial bones and cartilage. The dysregulation of cranial neural crest development patterns leads to congenital craniofacial disorders such as Craniosynostosis.
Understanding the mechanisms underlying neural crest development and growth could, in due course, grant patients the possibility of receiving regenerative therapies for tissues lost to several neurocristopathies.
This project brings together a variety of disciplines, integrating stem cell research, genomics and molecular biology, to identify novel craniosynostosis associated genes and design new tools to study neurocristopathies in vitro. We will make use of whole genome sequencing of over 250 patients, provided by the ErasmusMC Department of Pathology, as well as, the DECIPHER database to identify candidate gene variants causing neurocristopathies and place selected candidate genes in the different steps of Neural crest development.
Ultimately, we propose to: (1) Design hiPSC models from craniosynostosis patients to be used in disease modelling, mutation repairing, drug discovery and regenerative therapies; (2) Identify novel neurocristopathy related gene variants; (3) Contribute to the NEUcrest database focused on gene function in neurocristopathies.


ESR12 - Irina POVERENNAYA
Medical University of Vienna, Austria
I am Irina Poverennaya and my curiosity about applying single-cell technology led me to Prof. Dr. Igor Adameyko lab at the Medical University of Vienna where I started to work as a PhD student in September 2020. To get a Specialist's degree in bioengineering and bioinformatics from Lomonosov Moscow State University I had a comprehensive education in these and other biological fields that I would like to expand and apply to my PhD project.
It is important to be genuinely intrigued by the object of your study and with the neural crest it is very easy as this transient embryonic cell population is indeed full of wonder. Unique only to vertebrates they can migrate through the embryo and play a crucial role in the construction of a vertebrate body as they give rise to a broad range of different cell types including neurons, melanocytes, and bone cells, and provide an advanced peripheral nervous system, a cranial skeleton, improved endocrine regulation, dentition, etc. The induction of neural crest is intricately interlinked to the induction of neural tube (future central nervous system) and neurogenic placodes (future sensory organs) thus laying the foundation of the complex nervous system.
At the same time, neural crest cells might give rise to different types of human cancers, including pediatric and adulthood tumors of the sympathoadrenal system such as neuroblastomas, pheochromocytomas, and paragangliomas. Their intra- and inter-tumoral diversity might be defined by a plethora of the origin cell types - including the steps in the progression of a neural crest lineage in an embryo. The aim of my PhD project will be to characterize the neural crest lineage in human, mouse, and other model organisms with the help of spatial and single-cell transcriptomics in order to understand the genetic mechanisms of such progressions. The comparative analysis of the neural crest transitions and elucidation of the most conservative parts of GRNs and molecular modules will be performed in order to establish the regulatory cores that might be specifically important for tumor emergence and metastasis. The PhD project will also concern the evolution of such tumor-related circuits and the evolution of the neural crest in general. For this, the development of the early neural tube will be characterized in a lamprey, amphioxus, and saccoglossus animal models.

ESR13 - Nicolas XENIDIS
Tel Aviv University, Israel
I am Nicolas Xenidis, French PhD student at the Sackler School of Medicine – Tel Aviv University, under the supervision of Pr. Carmit Levy. Our lab is focused on melanocytes, with the goal to develop novel approaches in the prevention, diagnosis, and treatment of skin cancer in general and melanoma in particular.
During my academic years, I obtained a master’s degree in ‘Integrative Biology and Physiology’ at Sorbonne Université and completed my pharmacy degree specialized in research at Université Caen Normandie. My studies led to a strong interest in biomedical research focusing on the genetic and molecular foundation of pathologies with applied pharmacological and clinical research in order to improve the expectancy and quality of human life. My project stands at the interface between immunology and oncology.
Melanocytes are neural-crest derived dendritic cells located, inter alia, in the basal epidermis and producing melanin. Melanoma, a melanocyte originated neoplasm, is a very aggressive and treatment-resistant human skin cancer, with a number of 132,000 cases occurring globally each year. Causes are diverse and Ultra-Violet light exposure, by damaging melanocyte’s DNA, is a risk factor. Melanosomes are organelles storing the pigment melanin, produced by melanocytes and transported inside keratinocytes, where their main function is to protect cell DNA from UV light. This particular cell feature is conserved in melanoma with possible molecular changes. Findings from our lab uncovered how melanoma, through trafficking melanosomes, affects the formation of the dermal tumour niche and thus enhance invasion. Preliminary results are showing a close correlation between a highly pigmented melanoma phenotype and a low survival rate. Moreover, histopathological analysis of pigmented melanoma shows an important lymphocyte infiltration. However, Melanoma cells suppress the local immune response and survive. Understanding how pigmentation is involved in this inhibition will allow us to identify novel disease genes and thus develop novel therapeutic targets.
My project will focus on deciphering interactions between melanosomes and lymphocytes and on how melanosomes possibly act on lymphocytes activation or deactivation. Moreover, dissecting the underlying mechanisms of the molecular crosstalk between lymphocytes influenced by melanosomes would be a step more in understanding this immune evasion.

ESR14 - William BERTRANI TORRES
Institut Imagine, France
Hi, my name is William Bertani Torres and I am from São Paulo, Brazil! I have a bachelor's degree in Biomedical Sciences and a Master in Genetic counseling and human genomics. In 2020 I joined the NEUcrest network, working at the Imagine Institute, in Paris, France under the supervision of Drs. Bondurand and Pingault. Within the ITN, my PhD project's name and focus is "Cell and molecular bases of Waardenburg: exome sequencing, hiPSCs and NC-GRN".
Waardenburg syndrome (WS) is a rare disease characterized by the association of hearing loss and depigmentation abnormalities of hair, iris and skin. It is a neurocristopathy, which means that it affects the Neural Crest (NC) cells, melanocytes in particular, during development. These melanocytes are indeed responsible for pigmentation throughout the body and are also responsible for the maintenance of an electrolytic balance homeostasis in the organ of Corti, in the inner ear.
In our first objective, we are working with previous exome data of 16 patients, which all have been previously evaluated for pathogenic variants in the known genes of WS. Using different filtering strategies, we are trying to find candidate variants that can explain the phenotype and confirm the clinical diagnosis. We are also searching for genes of interest within online databases. After we find good candidate genes we would like to validate these genes by doing in vitro and in vivo functional analysis and determine the function of the proteins in melanocyte development.
In our second objective, we are working with previously established human induced Pluripotent Stem Cells (hiPSC) from patients carrying different SOX10 variants. SOX10 is one of the genes involved in type 2 WS, but also in more complex disorders affecting enteric nervous system, myelin of central and peripheral nervous system. For missense variants that lead to severe phenotype there is not yet a full explanation for it. So in objective 2 we will analyze the effect of different SOX10 variants on patient’s derived hiPSC differentiation capacities. With RNAseq we will study the deregulation of expression of genes, and compare these genes with the candidate genes from the exome analysis. This project will thus benefit fundamental research and may have relevance to diagnostic and medical care of the affected children.


ESR15 - Francesca LORENZINI
Azelead, France
I am Francesca Lorenzini and I started my PhD in the R&D division of AZELEAD, a biotechnology company based in Montpellier (France), in March 2020. I come from Italy, where I attended my Bachelor and my Master of Science at the University of Trento. I am a biotechnologist with a specialization in cancer biology. Since 2017 I have worked on zebrafish model for studying cancers. During my master internship I had the big opportunity to work on a project that ended with a publication: A network-based approach to identify deregulated pathways and drug effects in metabolic syndrome, by Misselbeck, Parolo, Lorenzini et al., Nature Commun, 2019, doi:10-1038/s-41467-019-13208-z. My current project within NEUcrest network focuses on the impact of tumour innervation by the peripheral nervous system both during the initiation phase of tumorigenesis and the dissemination of tumour cells throughout the body.
The peripheral nervous system (PNS) is one of the two components of the nervous system and it is derived from the neural crest (NC), a multipotent embryonic tissue. Over the years, different studies have demonstrated the capability of specific tumours cells of reactivating embryonic programs, like the epithelium-to-mesenchyme transition (EMT) of neural crest, during the early step of metastasis of NC-derived tumours (e.g. neuroblastoma and malignant melanoma). Thus, the final aim of my project is a better understanding of the metastasis process linked to tumour innervation by neural crest tissues allowing the final development of new drug candidates.
In order to reach this goal, I will use the zebrafish model, that allows the study of NC in the first phase of development and to perform an in vivo screening for the isolation of the best drug candidates able to modulate tumour innervation.
The first experiments will focus on the selection of candidate genes expressed in a specific branch of the PNS and on the study of the impact of these genes’ inactivation on tumour innervation. The final part of the work will be important for the identification of drug candidates.
The majority of my project will take place in the laboratories of AZELEAD, a biotechnology company based in Sanofi Campus in Montpellier (France). The team uses zebrafish model to study all the stages of metastatic progression with the aim to design novel compounds able to prevent metastasis and tumour progression in vivo. AZELEAD provides cutting edge technologies like high content imaging in 4D, high resolution imaging and platforms for pharmacological screening.
